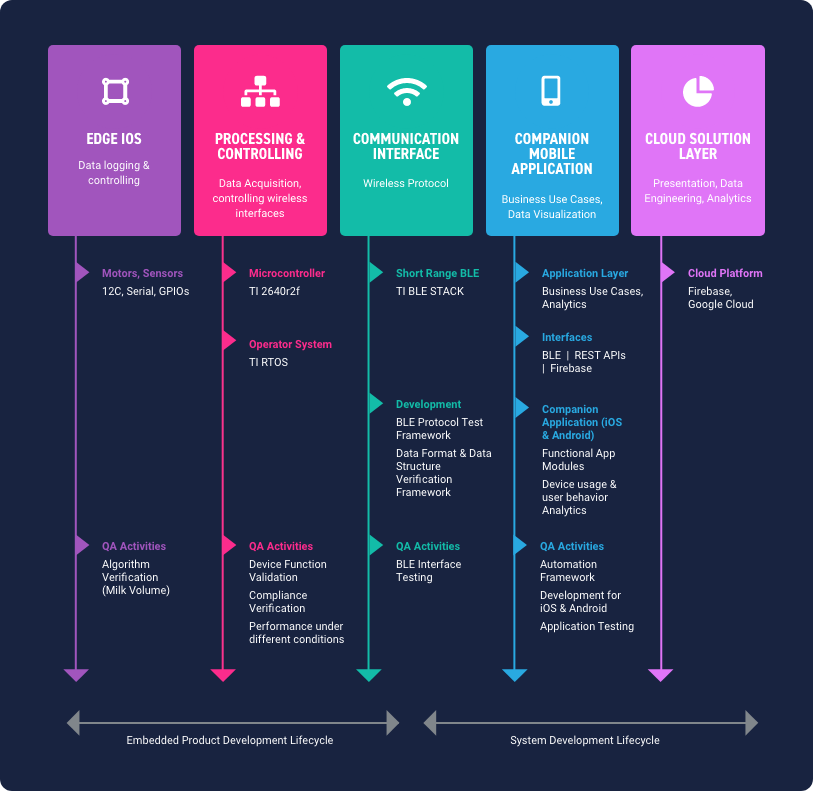
Willow, a Silicon Valley-based healthcare startup, is completely re-imagining the way busy moms pump breast milk with an innovative, patented technology called the Willow Wearable Breast Pump.
To realize its product vision and get it to market as quickly as possible, Willow required a combination of manufacturing innovation and digital technology expertise.